Here's a needle-in-a-haystack story with a happy ending. Leigh syndrome is a devastating genetic disease where the brain's power supply goes haywire. Patients develop severe, drug-resistant seizures that are incredibly difficult to treat. The brain has 86 billion neurons. How do you figure out which ones are actually causing the problem?
Researchers publishing in The Journal of Clinical Investigation did exactly that. They identified a very specific population of neurons that can't handle the energy crisis, and when those particular cells fail, seizures start. Better yet, they showed that fixing just that one circuit, not the whole brain, could prevent seizures and extend survival. Sometimes precision beats brute force.
The Energy Crisis in Your Head
Your mitochondria are the little power generators inside every cell, and your brain is their most demanding customer by far. Neurons are absolute energy hogs. They need constant power to maintain the electrical gradients that let them fire, to package and release neurotransmitters, to clean up afterward. It never stops.
In Leigh syndrome, a genetic mutation messes up the mitochondrial machinery. The power generators start failing. And while this affects cells throughout the body, the brain takes it especially hard because it's so energy-dependent.
But here's what had puzzled scientists: if mitochondria are malfunctioning everywhere in the brain, why do certain circuits crash first? Why seizures, specifically? The researchers behind this study wanted to find out which neurons were the weakest links in the energy crisis.
Finding the Vulnerable Population
To study this, the team worked with mice missing a gene called NDUFS4, which encodes a key component of the mitochondrial energy-production machinery. These mice develop Leigh syndrome and, tragically, fatal epilepsy. They're a good model for understanding what goes wrong in the human disease.
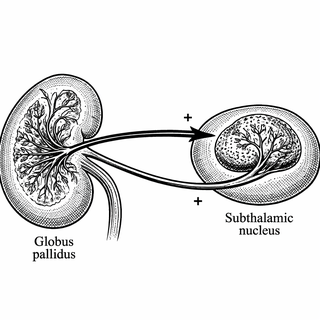
By carefully examining which neurons were most affected by the energy deficit, the researchers zeroed in on a very specific population: inhibitory neurons in a brain region called the external globus pallidus (GPe) that express a transcription factor called LHX6.
These neurons normally act like a brake pedal for another brain region called the subthalamic nucleus (STN). They keep the STN calm and under control. When these particular inhibitory neurons fail due to the energy crisis, they stop doing their job. The GPe loses its grip on the STN, and the STN starts misbehaving.
Without that brake? The subthalamic nucleus revs up, and seizures follow.
One Region, Not the Whole Brain
Here's where things get really exciting from a treatment perspective. The researchers used viral gene therapy to restore NDUFS4 expression, but they didn't try to fix the whole brain. They only targeted the GPe, that one specific region where the vulnerable neurons live.
The result was remarkable. Just restoring function to this one circuit prevented seizures and extended survival in the mice. They didn't need to rescue every affected cell in the brain. They just needed to fix the weak link.
This is a huge deal for thinking about therapy. Mitochondrial diseases have long been a therapeutic wasteland. We can diagnose them, but treatment options are limited. If you had to fix every cell in the brain, that's an almost impossible task. But if you only need to fix one circuit? That's suddenly much more tractable.
Another Angle: Calm the Overexcited Region Directly
The researchers also tried another approach. Instead of fixing the failing inhibitory neurons, what if you just directly calm down the subthalamic nucleus that's become overactive?
That worked too. Reducing STN activity reduced seizures.
This finding is particularly interesting because the STN is already a target for deep brain stimulation in other conditions, most notably Parkinson's disease. We already have clinical tools and experience with modulating this exact brain region. The infrastructure exists.
Could DBS or other STN-targeting approaches help patients with Leigh syndrome? It's an open question that now makes sense to ask. The basic science has pointed toward a treatment strategy that leverages technology we already have.
What This Means for Mitochondrial Disease
For years, the thinking about mitochondrial diseases was somewhat fatalistic. The mutation is everywhere. The dysfunction is everywhere. How could you possibly treat it? You can't replace every mitochondrion in the body.
This research offers a different perspective. Yes, the mutation is everywhere. But the clinical symptoms come from specific vulnerable circuits. If you can identify those circuits, you might be able to target them specifically and get outsized therapeutic benefit.
It's a proof of concept that precision can win even when the underlying disease is global. Find the cells that matter most for the symptoms you're trying to prevent. Focus your therapeutic firepower there.
A Reason for Optimism
Leigh syndrome remains a devastating disease with limited treatment options. Nothing in this paper changes that for patients today. But it does change the trajectory of the research.
Scientists now have a clear target: the LHX6-expressing neurons in the GPe and their connections to the STN. They have proof that rescuing this circuit prevents seizures. And they have potential therapeutic approaches, including gene therapy and neuromodulation techniques, that could be developed further.
From 86 billion neurons to a handful of critical cells. That's the power of figuring out which circuit actually matters.
Reference: Quintana A, et al. (2025). Dysfunctional LHX6 pallido-subthalamic projections mediate epileptic events in a mouse model of Leigh syndrome. The Journal of Clinical Investigation. doi: 10.1172/JCI187571 | PMID: 41321311
Disclaimer: The image accompanying this article is for illustrative purposes only and does not depict actual experimental results, data, or biological mechanisms.
