Somewhere inside you right now, a protein the size of a molecular freight train is quietly hauling fat from one membrane to another. VPS13A clocks in at over 3,000 amino acids - making most other proteins look like a text message by comparison - and it works by forming a greased-up tunnel between your cell's ER and plasma membrane, sliding lipids through like some kind of biological Slip 'N Slide.
When it works? You'd never notice. When it breaks? Your neurons start dying and your red blood cells go full sea urchin. Scientists just figured out exactly how patient mutations wreck this thing, and the answer came in four surprisingly neat categories - with one genuinely wild plot twist (Lin et al., 2026).
The World's Most Consequential Handshake
VPS13A doesn't run its lipid delivery service solo. It partners up with a membrane protein called XK, and together they pull off phospholipid scrambling - the controlled flipping of fat molecules across your cell membrane when ATP comes knocking. Picture a revolving door for lipids: VPS13A feeds it, XK spins it. Break either half and the whole operation collapses.
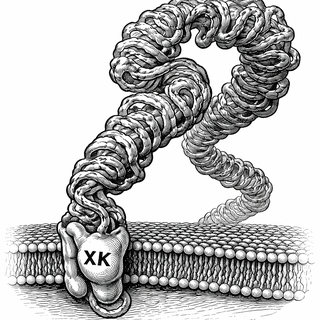
That collapse has a name: VPS13A disease (formerly chorea-acanthocytosis). It's rare - maybe 500 to 1,000 people worldwide - and it's merciless. It typically shows up around age 30 with involuntary movements, seizures, cognitive decline, and those distinctive spiky red blood cells that pathologists call acanthocytes (GeneReviews, NCBI). There's no cure, no targeted treatment, and until now, a frustrating lack of clarity on what patient mutations actually do at the molecular level.
The Nagata lab has been chipping away at this problem for years. Their earlier work showed that XK acts as a lipid scramblase while VPS13A works as the lipid supplier, and that the two proteins physically grab onto each other to coordinate the whole operation (Ryoden & Nagata, 2022). But precisely how they bind - and precisely how patient mutations sabotage that binding - was still an open question.
A Mutation Field Guide (With One Surprise Villain)
The team built a mouse cell system expressing full-length VPS13A (which, given this protein is basically an aircraft carrier, deserves applause on its own) and tested ten mutations found in real patients. What they found was unexpectedly tidy - four distinct modes of failure:
The No-Shows (L67P, I90K, W2453R): These mutations tank VPS13A expression. The protein gets built but can't fold properly, so the cell's quality control shreds it before it can do anything. No protein, no service. Like ordering a pizza that never leaves the kitchen.
The Warm Bodies (A1091P, M3080R): Normal protein levels, zero scramblase activity. The protein clocks in, sits at its desk, and stares at the wall. Present but spectacularly useless.
The Walking Wounded (S1446P, Q2689H, Y2713C, R3084H): These modestly impair either expression or activity. Not an immediate catastrophe, but enough of a slow leak that over decades, things fall apart. Death by a thousand paper cuts, neuron-style.
The Rogue Agent (I2763R): Here's where it gets weird. Instead of just losing function like a well-behaved disease mutation, this one gained a new trick nobody asked for. It actively changed cell size and disrupted the ER - and did it completely independently of XK. That's not a broken machine. That's a machine that started freelancing.
Why the Rogue Agent Changes Everything
VPS13A disease has been treated as a loss-of-function story: protein breaks, lipids can't move, neurons die. Finding a gain-of-function mutation means at least some patients are dealing with a fundamentally different problem. Their cells aren't just missing a service - they're being actively sabotaged by a protein gone rogue. For anyone trying to develop therapies, that's the difference between replacing a missing part and stopping a machine from doing damage.
The Molecular Blueprint
The team also mapped out exactly where VPS13A grabs XK: a C-terminal beta-strand on VPS13A slots into a beta-hairpin in XK's midsection. Break that handshake and scrambling dies completely. They even showed that XKR2, a cousin of XK with a similar structural motif, can step in as a substitute partner and support scrambling. The protein is apparently less monogamous than we assumed - it just needs the right grip.
These structural details matter because VPS13A belongs to a family of bridge-like lipid transporters now linked to conditions from early-onset Parkinson's to developmental disorders (Hanna et al., 2023; Swan, 2025). Understanding how one member of this family breaks gives us a playbook for the whole clan.
For a disease with no approved treatment, knowing mutations break things in four fundamentally different ways isn't just neat science. It's the roadmap to precision therapies that treat the actual problem instead of guessing. The lipid delivery service is down, but at least now we know which parts of the truck to fix.
References
-
Lin, X., Ryoden, Y., Suzuki, C., Ishikawa, H., Sakuragi, T., Uchiyama, Y., & Nagata, S. (2026). Four subtypes of disease-causing missense mutations underlie pathogenic protein interactions in neurodegenerative VPS13A disease. The Journal of Clinical Investigation. DOI: 10.1172/JCI200890. PMID: 41874565
-
Guillén-Samander, A., Wu, Y., Pineda, S. S., García, F. J., Eisen, J. N., Leonzino, M., Ugur, B., Kellis, M., Heiman, M., & De Camilli, P. (2022). A partnership between the lipid scramblase XK and the lipid transfer protein VPS13A at the plasma membrane. Proceedings of the National Academy of Sciences, 119(35), e2205425119. DOI: 10.1073/pnas.2205425119. PMCID: PMC9436381
-
Ryoden, Y., & Nagata, S. (2022). The XK plasma membrane scramblase and the VPS13A cytosolic lipid transporter for ATP-induced cell death. BioEssays, 44(10), e2200106. DOI: 10.1002/bies.202200106. PMID: 35996795
-
Hanna, M., Guillén-Samander, A., & De Camilli, P. (2023). RBG motif bridge-like lipid transport proteins: Structure, functions, and open questions. Annual Review of Cell and Developmental Biology, 39, 409-434. DOI: 10.1146/annurev-cellbio-120420-014634
-
Swan, L. E. (2025). VPS13 and bridge-like lipid transporters, mechanisms, and mysteries. Frontiers in Neuroscience, 19, 1534061. DOI: 10.3389/fnins.2025.1534061. PMCID: PMC12066543
Disclaimer: The image accompanying this article is for illustrative purposes only and does not depict actual experimental results, data, or biological mechanisms.
