Ten years ago, we mostly talked about chronic pain like it was one busted alarm system that just would not shut up. Now the picture looks messier and much more interesting - more like a badly run restaurant where one cook burns the food while another starts an argument in the dining room. Sorry, that is a terrible metaphor, and I regret it immediately, but this new mouse study gives it some real bite.
Researchers looked at the medial shell of the nucleus accumbens, a small brain region better known for reward, motivation, and all the stuff that makes life feel worth putting pants on for. In male mice with chronic neuropathic pain and depression-like behavior, they found that two neighboring cell populations were both quieter than usual. But when the team nudged them back on, they did different jobs. D1 neurons mainly improved depression-like behavior. D2 neurons mainly reduced pain. Same neighborhood, different employees, very different shift duties. [1]
The Brain’s Complaint Department
This matters because chronic pain and depression love to travel as a terrible duo. Pain can flatten motivation, shrink pleasure, and make everything feel like walking through wet cement. Depression, in return, can crank up pain sensitivity and make coping harder. It is less a neat cause-and-effect chain and more a miserable slow-cooker recipe where each ingredient keeps intensifying the other. Reviews over the last few years have made the same point: chronic pain is not just a sensation problem. It also recruits emotion, motivation, and decision-making circuits. [2,3]
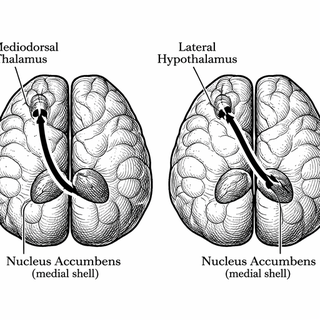
The nucleus accumbens sits right in the middle of that mess. It is part of the ventral striatum, packed with medium spiny neurons that act like tiny gatekeepers for motivation and action selection. Wikipedia is useful here for the quick refresher version: the accumbens shell helps process reward, aversion, and behavioral drive, while its D1- and D2-type neurons often contribute differently to output pathways. Translation: this is not just a “feel good” blob. It is a traffic circle where pain, mood, and motivation all show up honking.
Two Routes Out of the Same Weird Neighborhood
The clever bit in this paper is that the authors did not stop at “these cells change.” They traced where those cells send signals.
When they activated D1 medium spiny neurons in the medial accumbens shell, the biggest effect was on depression-like behavior. That pathway projected to the mediodorsal thalamus, a region heavily tied to cognition and affect. When they activated D2 neurons, the mice got pain relief instead, and that pathway projected to the lateral hypothalamus. So the study sketches a split arrangement: one route seems more involved in the emotional misery that often comes with chronic pain, while another route seems more involved in the pain itself. [1]
That is a useful upgrade from the older, fuzzier idea that the accumbens just sort of “modulates suffering” in a broad hand-wavy way. Recent work has been pushing the field toward finer circuit maps. A 2023 Nature Communications paper also found separable circuits contributing to chronic pain-like and depression-like behaviors in male mice, though through ventral pallidal cholinergic pathways rather than the exact accumbens outputs highlighted here. [4] A 2025 JCI paper went even further, showing that distinct medial accumbens shell pathways can drive different symptom clusters in this comorbidity model. [5] The trend is hard to miss: the brain is not using one giant misery lever. It is using multiple knobs, because of course it is.
Why You Should Care Even If You Are Not a Mouse
If these findings hold up across labs, sexes, and eventually humans, they hint at a better way to think about treatment. Right now, chronic pain and depression often get treated like two separate dishes on the menu. One specialist handles the pain. Another handles the mood. Meanwhile the brain is back there reducing both sauces in the same pan until everything tastes burnt.
This study suggests symptom-specific targeting might be possible. Maybe future therapies could calm the pain-heavy circuit without flattening mood, or boost the mood-related circuit without messing with sensory processing. That could mean more precise brain stimulation, better drug targets, or biomarker-guided treatments that match the dominant problem in a given patient. There is already human work linking nucleus accumbens connectivity with chronic pain risk and persistence, so the translational bridge is not pure science fiction anymore. [3]
Still, nobody should sprint from this paper to “amazing, we solved chronic pain.” These were male mice. The behavioral measures are informative but not identical to human pain or human depression. And circuit-level interventions in a lab are much cleaner than anything a clinic can do on a Wednesday between insurance calls.
Even so, the core idea is strong: chronic pain and its emotional side effects may share a kitchen, but they do not come from the exact same burner. That is good news, because if you want to fix a bad meal, it helps to know whether the soup is too salty or the stove is on fire. Another terrible cooking joke, yes. I know. The brain made me do it.
References
- Xia XX, Wang YH, Wang XY, Liu XQ, Hu W, Liu XF, Zhang Y. The medial shell of nucleus accumbens regulates chronic pain and comorbid depression via separate downstream targets in male mice. Cell Reports. 2025;45(1):116716. DOI: https://doi.org/10.1016/j.celrep.2025.116716
- Lindsay NM, Chen C, Gilam G, Mackey S, Scherrer G. Brain circuits for pain and its treatment. Science Translational Medicine. 2021;13(619):eabj7360. DOI: https://doi.org/10.1126/scitranslmed.abj7360 PMCID: https://pmc.ncbi.nlm.nih.gov/articles/PMC8675872/
- Sunavsky A, Hashmi MA, Robertson JW, Veinot J, Hashmi JA. The nucleus accumbens-prefrontal connectivity as a predictor of chronic low back pain. Pain. 2025;166(10):e363-e377. DOI: https://doi.org/10.1097/j.pain.0000000000003620
- Ji YW, Shen ZL, Zhang X, Zhang K, Jia T, Xu X, Geng H, Han Y, Yin C, Yang JJ, Cao JL, Zhou C, Xiao C. Plasticity in ventral pallidal cholinergic neuron-derived circuits contributes to comorbid chronic pain-like and depression-like behaviour in male mice. Nature Communications. 2023;14(1):2182. DOI: https://doi.org/10.1038/s41467-023-37968-x
- Liu D, Xu FX, Yu Z, Huang XJ, Zhu YB, Wang LJ, Wu CW, Zhang X, Cao JL, Li J. Distinct nucleus accumbens neural pathways underlie separate behavioral features of chronic pain and comorbid depression. Journal of Clinical Investigation. 2025;135(15):e191270. DOI: https://doi.org/10.1172/JCI191270 PMCID: https://pmc.ncbi.nlm.nih.gov/articles/PMC12321395/
Disclaimer: The image accompanying this article is for illustrative purposes only and does not depict actual experimental results, data, or biological mechanisms.
