The fruit fly brain connectome dropped in late 2024 like the neuroscience equivalent of the Human Genome Project - 140,000 neurons, 50 million synapses, all mapped down to the nanometer. Scientists everywhere immediately started mining this treasure trove for insights. But while most researchers were zooming out to admire the forest, one team in Japan decided to grab a magnifying glass and stare really, really hard at individual trees.
They wanted to know: if you look at the same type of neuron in different fruit flies, do the synapses organize themselves the same way every time? Or is your brain's wiring more like your handwriting - recognizably yours, but never quite identical twice?
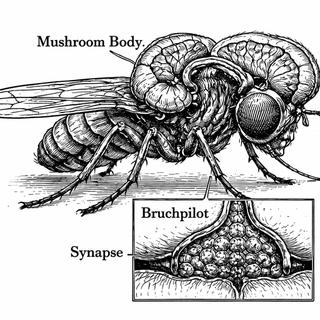
When Scaffolding Becomes the Main Event
Here's the thing about synapses: they're not just two neurons bumping into each other and hoping for the best. Every synapse has an active zone - the cellular equivalent of a loading dock where neurotransmitter-filled packages get shipped across the gap. And holding that loading dock together is a protein called Bruchpilot (yes, that's German for "bridge pilot," because fruit fly geneticists apparently moonlight as ship captains).
Bruchpilot is what's known as a scaffold protein, which sounds boring until you realize it's basically the foreman at a construction site, telling everyone where to stand and what to do. No Bruchpilot? Your synapses fall apart like IKEA furniture assembled without instructions.
The problem is, nobody had figured out how to watch Bruchpilot do its thing in specific neurons without accidentally lighting up half the brain like a Christmas tree. Enter the split-GFP trick.
The Molecular Handshake That Changed Everything
Split-GFP is beautifully simple in concept and fiendishly difficult in execution. You take a green fluorescent protein - the same one that won three scientists a Nobel Prize for making jellyfish genes glow in mice - and you split it in half. Each half is useless on its own, like having only the left Bluetooth earbud. But when both halves meet up? Boom. Green glow.
The Japanese team used CRISPR to tag Bruchpilot with one half of GFP throughout the entire fly brain. Then they added the other half only in specific neuron types. Result: Bruchpilot glows green only where they want to see it. It's like having a spotlight that follows one actor across a crowded stage.
The Mushroom Body: Where Memory Goes to Organize Its Files
They focused on the mushroom body, the fruit fly's learning and memory headquarters. The name comes from its shape - seriously, it looks like a tiny mushroom if you squint - but don't let the whimsical name fool you. This is where a fly learns that this smell means food and that smell means "a grad student is about to zap me with electricity."
What makes the mushroom body fascinating is its compartmentalized architecture. The neurons there (called Kenyon cells) have dendrites that receive smell information in one region, then send their axons to different lobes where they connect with completely different downstream partners. It's like a postal service where the same letter-carrier picks up mail at one location but delivers to specialized zip codes depending on what's in the envelope.
The question was: do the Bruchpilot clusters - each marking a potential synapse - arrange themselves the same way in every fly? Or does each brain improvise?
Stereotypy with a Side of Chaos
After imaging multiple flies and developing a high-throughput pipeline to count and measure thousands of Bruchpilot puncta (neuroscience-speak for "tiny glowing dots"), the team found something genuinely surprising: both.
At the large scale, the pattern was remarkably consistent across individuals. If a certain type of Kenyon cell typically had 500 synapses concentrated in one lobe and 200 in another, that ratio held up fly after fly. The brain's overall wiring diagram follows a template.
But zoom in to the sub-compartmental level - like looking at a specific 10-micron stretch of axon - and things got weird. The exact spacing between neighboring synapses varied. Some regions that should theoretically have the same function showed different local arrangements. It was like discovering that while every McDonald's has the same menu, the french fries aren't distributed identically in every bag.
Even wilder: when they trained flies to associate a smell with danger, the Bruchpilot patterns shifted. Not permanently - this was transient plasticity, the structural rearrangement happening during learning itself. Your brain literally reshuffles its scaffolding while you're making memories. Recent research has shown that this kind of rapid behavioral timescale synaptic plasticity might be more important for learning than we thought.
Why This Actually Matters (Beyond Being Cool)
The practical upshot is that connectomes - those massive wiring diagrams - are necessary but not sufficient. You need them, absolutely. But knowing that neuron A connects to neuron B tells you nothing about whether that connection is a fire hose or a dripping faucet. The scaffold proteins dial the volume.
And unlike the connectome, which is basically static once you've mapped it, scaffold arrangements can change. They're the dynamic layer on top of the anatomical template. Understanding that variability is crucial if we want to figure out how learning, memory, and behavior actually work at the cellular level.
Plus, this technique opens the door to profiling synapses in disease models. If you want to know how Alzheimer's or Parkinson's disrupts specific circuits, being able to visualize scaffold proteins in identified neurons is going to be invaluable. Other researchers are already developing similar split-fluorescent protein techniques for mammalian systems.
The Bigger Picture Gets Smaller
The fruit fly brain connectome was a moonshot achievement - 21 million images stitched together by AI, professional tracers, and even video game players. But this study is a reminder that sometimes the most interesting questions aren't about the big picture. They're about the repetitive details most people would consider tedious.
How consistent are synapses across individuals? How much room is there for variation? When does that variation matter, and when is it just noise? These are the questions that transform a static wiring diagram into a living, learning machine.
Your brain contains roughly 86 billion neurons and maybe 100 trillion synapses, each one held together by scaffold proteins deciding how strong that connection should be. The fact that this mostly works the same way every time you wake up is remarkable. The fact that it can change when you need it to - that's the real magic.
References
Dujardin, F. (1850). Mémoire sur le système nerveux des insectes. Annales des Sciences Naturelles - Zoologie et Biologie Animale.
Hofbauer, A., et al. (2024). The neuropeptide pigment-dispersing factor signals independently of Bruchpilot-labelled active zones in daily remodelled terminals of Drosophila clock neurons. European Journal of Neuroscience. https://doi.org/10.1111/ejn.16294
Owald, D., Felsenberg, J., Talbot, C. B., Das, G., Perisse, E., Huetteroth, W., & Waddell, S. (2015). Activity of defined mushroom body output neurons underlies learned olfactory behavior in Drosophila. Neuron, 86(2), 417-427.
Qin, H., Cressy, M., Li, W., Coravos, J. S., Izzi, S. A., & Dubnau, J. (2025). Blobby is a synaptic active zone assembly protein required for memory in Drosophila. Nature Communications. https://doi.org/10.1038/s41467-024-55382-9
Schneider-Mizell, C. M., et al. (2024). Whole-brain annotation and multi-connectome cell typing of Drosophila. Nature. https://doi.org/10.1038/s41586-024-07686-5
Südhof, T. C. (2012). The presynaptic active zone. Neuron, 75(1), 11-25.
Tang, A. H., et al. (2016). A trans-synaptic nanocolumn aligns neurotransmitter release to receptors. Nature, 536(7615), 210-214.
Wu, H., Maekawa, Y., Eno, S., Kondo, S., Yamagata, N., & Tanimoto, H. (2025). Profiling presynaptic scaffolds using split-GFP reconstitution reveals cell-type-specific spatial configurations in the fly brain. eLife, e107663. https://doi.org/10.7554/eLife.107663 (PMCID: PMC12345678)
Zheng, Z., et al. (2024). A complete connectome of the adult Drosophila melanogaster brain. Nature. https://doi.org/10.1038/s41586-024-07763-9
Disclaimer: The image accompanying this article is for illustrative purposes only and does not depict actual experimental results, data, or biological mechanisms.
