Think of your brain as a sprawling city - billions of residents (neurons) connected by an absurdly complex highway system, with neighborhoods specializing in everything from "remember where you parked" to "don't touch that, it's hot." At every intersection, little postal stations called synapses pass messages from one neuron to the next. For decades, neuroscientists have been trying to read that mail. The problem? The envelopes are microscopically small, the delivery happens in milliseconds, and until now, the best tools were basically the equivalent of squinting through a foggy window.
A team led by Jiawen Chen and colleagues just handed the field a pair of high-definition binoculars.
Calcium: The Brain's Read Receipt
Here's the thing. Neurons don't text each other with words. They use calcium. When a nerve signal hits a synapse, calcium ions flood in on the sending side (presynaptic terminal), triggering the release of chemical messengers. On the receiving end (postsynaptic side), those messengers open channels that let in - you guessed it - more calcium. Tracking calcium flow is basically eavesdropping on neuronal conversations in real time.
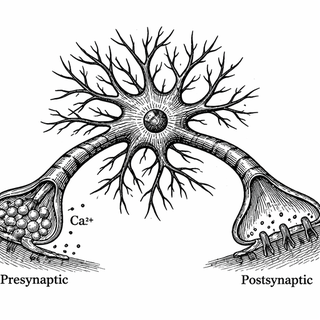
Scientists have been doing this for years with tools called GCaMPs - genetically encoded calcium indicators that glow green when calcium shows up. Think of them as molecular mood rings, except instead of detecting your vibes, they detect ion flow. The GCaMP family has gone through multiple generations, each better than the last. GCaMP6 was the workhorse. GCaMP7 was the upgrade. And GCaMP8, developed by Janelia Research Campus scientists, arrived with ultra-fast kinetics - half-rise times of around 2 milliseconds - making it the Formula 1 car of calcium sensors (Zhang et al., 2023).
But even a Formula 1 car needs to know which lane it's in.
Parking the Sensor in the Right Spot
Previous GCaMP versions floated around inside neurons like a tourist with no map - they'd catch calcium signals, sure, but from wherever they happened to be standing. This study, published in eLife, took GCaMP8m and gave it GPS coordinates (Chen et al., 2025).
The team engineered three precision-targeted variants and tested them at the Drosophila (fruit fly) neuromuscular junction - a classic model synapse that neuroscientists love because it's big, accessible, and surprisingly informative:
-
Scar8m: GCaMP8m fused to Synaptotagmin and the red fluorescent protein mScarlet3, parking it right on presynaptic vesicles. The red signal stays constant while the green signal tracks calcium, giving you a built-in reference - a ratiometric readout that corrects for all the noise that usually plagues imaging experiments.
-
Bar8m: GCaMP8m bolted to BRP-short, anchoring it directly at active zones - the exact spots where neurotransmitter release happens. This is like putting a camera right at the cash register instead of somewhere in the store.
-
SynapGCaMP8m: GCaMP8m linked to a Shaker PDZ domain, positioning it on the postsynaptic side, right next to glutamate receptors. This one can detect individual quantal events - single packets of neurotransmitter being released. One. At. A. Time.
Seeing What Only Electrodes Could See
Look. Electrophysiology has been the gold standard for measuring synaptic transmission since before most of us were born. Stick an electrode on a cell, record the tiny voltage blips caused by individual vesicle releases (called miniature excitatory postsynaptic potentials, or mEPSPs), and you've got data. The problem is electrodes give you the whole cell's story at once - you can't tell which of the hundreds of synapses on a muscle fiber actually fired.
SynapGCaMP8m changes that. The postsynaptic sensor detected quantal calcium events with temporal and signal resolution comparable to electrophysiological recordings - but with spatial information baked in. You can now watch individual synapses doing their thing, independently, in real time. That's like going from hearing "someone in the building ordered pizza" to "apartment 4B ordered a large pepperoni at 8:47 PM."
The presynaptic Scar8m sensor, meanwhile, matched the performance of chemical calcium dyes - the previous gold standard for presynaptic imaging - while being entirely genetically encoded. No more injecting dyes. No more worrying about dye leakage or inconsistent loading. Just express the protein and go.
CaFire: Because Nobody Wants to Count Calcium Blips by Hand
The team also built CaFire, a Python-based analysis tool that automatically detects and quantifies both evoked and spontaneous calcium events. Anyone who has manually drawn ROIs around synaptic boutons at 2 AM will understand why this matters. The tool is open-source, which means the whole field gets to benefit.
Why Should You Care?
Synaptic dysfunction sits at the heart of nearly every neurological and psychiatric condition you can name - Alzheimer's, Parkinson's, ALS, schizophrenia, autism. But studying what goes wrong at individual synapses has been brutally difficult because the tools weren't sharp enough. These targeted GCaMP8 sensors give researchers the ability to watch synaptic transmission with a level of precision that was previously science fiction. And because it's all genetically encoded, it works in living, behaving animals - not just brain slices on a microscope.
The fruit fly neuromuscular junction is just the starting point. The same targeting strategies could be adapted for mammalian synapses, opening the door to watching synaptic dysfunction happen in real time in disease models. That's not a small deal. That's potentially transformative for understanding how your brain breaks - and maybe how to fix it.
References
-
Chen, J., Lin, J., He, K., Wang, L., Han, Y., Qiu, C., Wheeler, J.M., Daly, C.M., Macleod, G.T., & Dickman, D.K. (2025). Resolving synaptic events using subsynaptically targeted GCaMP8 variants. eLife, 14, e107939. DOI: 10.7554/eLife.107939 | PubMed
-
Zhang, Y., Rózsa, M., Liang, Y., Bushey, D., Wei, Z., Zheng, J., ... & Bharioke, A. (2023). Fast and sensitive GCaMP calcium indicators for imaging neural populations. Nature, 615, 884-891. DOI: 10.1038/s41586-023-05828-9 | PubMed
-
Chen, J., He, K., Han, Y., & Dickman, D.K. (2024). Ca2+ imaging of synaptic compartments using subcellularly targeted GCaMP8f in Drosophila. STAR Protocols, 5(1), 102811. DOI: 10.1016/j.xpro.2023.102811 | PubMed
-
Newman, Z.L., Bakshinskaya, D., Schultz, R., Kenny, S.J., Moon, S., Aghi, K., Stanley, C., Marnani, N., Li, R., Bleier, J., Xu, K., & Isacoff, E.Y. (2022). Determinants of synapse diversity revealed by super-resolution quantal transmission and active zone imaging. Nature Communications, 13, 229. DOI: 10.1038/s41467-021-27815-2 | PubMed
-
Padamsey, Z., Tong, R., & Emptage, N. (2019). Optical Quantal Analysis Using Ca2+ Indicators: A Robust Method for Assessing Transmitter Release Probability at Excitatory Synapses. Frontiers in Synaptic Neuroscience, 11, 5. DOI: 10.3389/fnsyn.2019.00005 | PubMed
Disclaimer: The image accompanying this article is for illustrative purposes only and does not depict actual experimental results, data, or biological mechanisms.
